Quiz of the month
A 69-year-old man with a history of liver transplant for hepatitis C–related cirrhosis and hepatocellular carcinoma in 2010 on cyclosporine, mycophenolate, and prednisone, mantle cell lymphoma on ibrutinib, pancreatic cancer status post resection in 2009 CKD stage 4; and hypertension presents with right arm pain and swelling, dysuria, and shortness of breath requiring 3 L/min nasal cannula. He reports a two-week history of cough with difficulty expectorating and intermittent wheezing. He denies fever, chills, hemoptysis, orthopnea, lower-extremity edema, or night sweats. On exam, he is lethargic , afebrile. He is hemodynamically stable. Lungs are clear to auscultation, there is no palpable adenopathy. Right arm and elbow is markedly edematous and erythematous, with fluctuance, no crepitus, no purulence or drainage. Blood cultures are negative, and echocardiography without vegetations. Complete blood count with mild anemia and normal WBC.
The following are representative cuts of his CT chest.
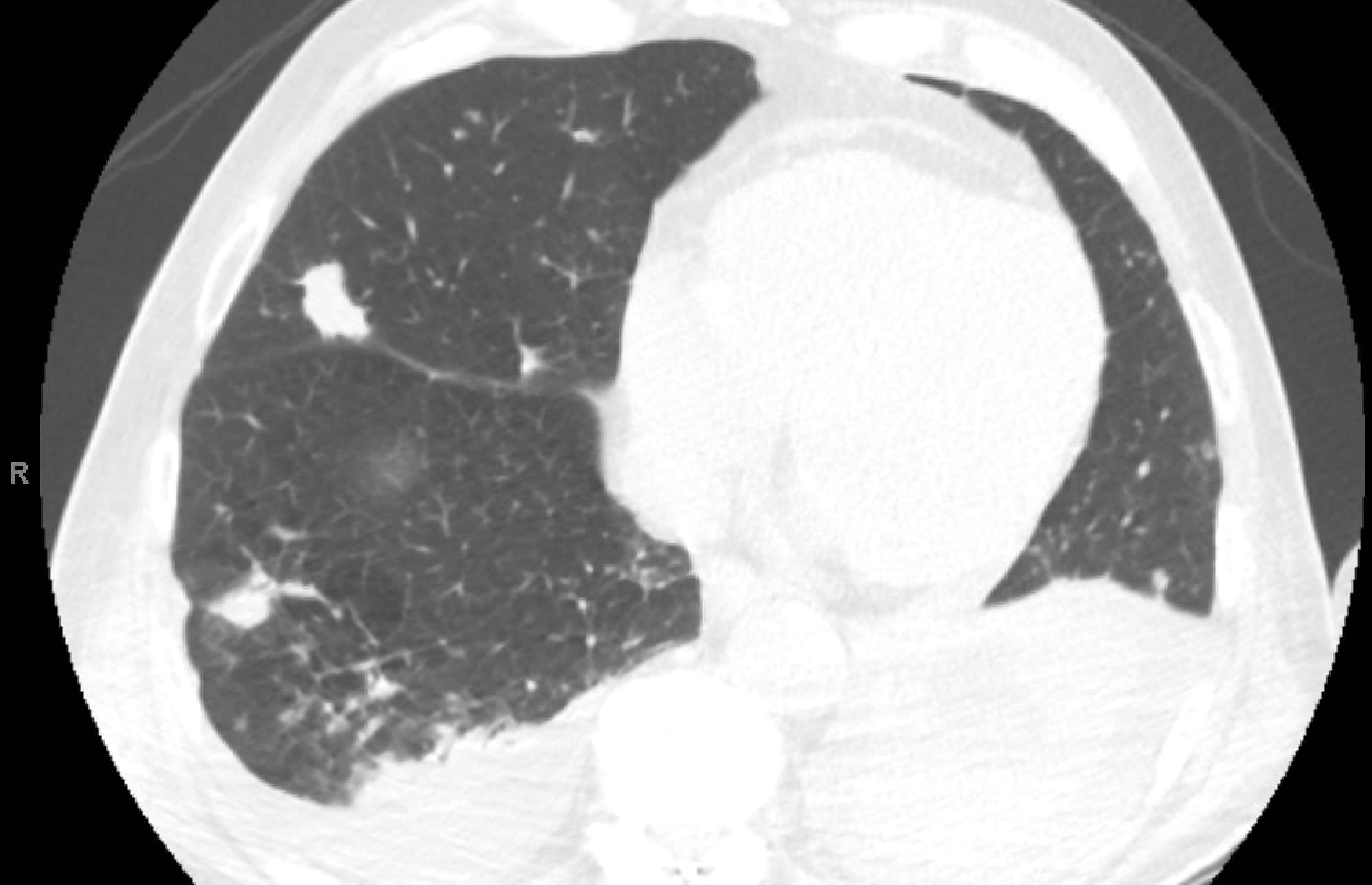
 Which of the following is the most plausible diagnostic consideration?
Which of the following is the most plausible diagnostic consideration?
A. Septic pulmonary emboli
B. Pulmonary nocardiosis
C. Invasive aspergillosis
D. Pulmonary metastases
E. Reactivation tuberculosis
Answer to November quiz of the month:
D. Vanishing Lung Syndrome
Vanishing lung syndrome, also known as idiopathic giant bullous emphysema, is a disease first described 1937, characterized by giant bulla that occupies at least one third of one or both lungs, causing compression on the surrounding normal lung parenchyma, creating the appearance of “disappearing lung” on imaging. The disease is frequently asymmetrical. It typically affects young men with a longstanding history of smoking (Ladizinski & Sankey, 2014) and rarely occurs in nonsmokers. Association with alpha-1 antitrypsin deficiency and marijuana use has been described, with a proposed pathophysiological mechanism alveolar wall destruction due to elastase (Talwar at al., 2022).
Clinical presentation usually includes cough, dyspnea, in some cases chest pain due to increasing size of the bullae, pneumothorax from bullae rupture but is occasionally incidentally detected in asymptomatic individuals.
Chest xray is usually the initial investigation and may reveal asymmetric hyperlucent lungs. HRCT images confirm the diagnosis with frequent findings of multiple large bullae in both lungs, which range in size from few centimeters to very large dimensions, almost filling the entire lung. Occasionally, it may be difficult to differentiate between a large bulla and a pneumothorax. A double-wall sign (air seen on both sides of the bulla wall) may be used to differentiate between the two conditions (Waitches, 2000).
Complications include secondary infection of the bullae with aging, finding of an air fluid level, and rupture of bullae leading to pneumothorax.
Asymptomatic cases are managed conservatively. Lung-volume–reduction surgery is considered for selected symptomatic patients with bullectomy.
References:
Ladizinski, B, M.D., Sankey, C. (2014). Vanishing Lung syndrome. N Engl J Med 2014;370: e14. https://www.nejm.org/doi/10.1056/NEJMicm1305898
Talwar, D., Andhale, A., Acharya, S., Kumar, S., & Talwar, D. (2022). Vanishing lung syndrome masquerading as pneumothorax in a smoker: Now you see me, now you do not. Lung India : official organ of Indian Chest Society, 39(4), 374–376. https://doi.org/10.4103/lungindia.lungindia_715_21
Waitches, G. M., Stern, E. J., & Dubinsky, T. J. (2000). Usefulness of the double-wall sign in detecting pneumothorax in patients with giant bullous emphysema. AJR. American journal of roentgenology, 174(6), 1765–1768. https://doi.org/10.2214/ajr.174.6.1741765