Please also see syllabus
Overview
Completion of scholarly work is a core requirement of the fellowship, with the primary expectation being a fellow-initiated original research project that defines and tests a clear hypothesis. In some cases, a rigorously designed quality improvement initiative may also meet this requirement, particularly when it evaluates a defined process with a testable hypothesis. Case reports represent an additional, but secondary, form of scholarly activity.
Participation in original research develops the important skill set of understanding the concept of the scientific method (asking a question, developing a hypothesis, testing the hypothesis, and drawing conclusions from the testing).
Requirement # 1: Project of scholarly activity
- Fellow must be the primary driver of the project
- Must present the work at a national conference
- Project must be complete and in written form before graduation
Fellow can choose from one of these categories
1. Original research
2. Quality Initiative project (Must be the primary fellow, as defined by the person presenting at a conference, and the primary author of the paper)
4. Review article if submitted to a peer-reviewed journal.
Requirement # 2: Research curriculum
Each research elective includes assigned readings, lectures, and required tasks. These materials are available on the Research Curriculum page at hfhpulm.com. Content is organized by year of fellowship and is intended to build progressively. Fellows are expected to complete the assigned items before advancing to the next phase of training.
Requirement # 3: Presenting original project at a national meeting
Fellows must present their original research project at a national conference. This must involve presentation of findings from a fellow-initiated project with a defined hypothesis and analysis. Case reports do not meet this requirement.
Acceptable venues include national meetings such as American College of Chest Physicians (CHEST), American Thoracic Society (ATS), Society of Critical Care Medicine (SCCM), or other comparable national conferences.
Ideas for research projects
1. Abandoned or Unfinished Projects
Fellows may choose to continue a prior project that was not completed by a graduating fellow. These projects are available for review in the Research section of hfhpulm. You are free to select from this list and advance the work to completion.
2. Directed Reading
Review the current literature to identify gaps in knowledge, unresolved clinical questions, or areas where methodology could be improved. A focused review often leads naturally to a testable hypothesis.
3. Discussion with a Mentor
Meet with faculty mentors to refine ideas, assess feasibility, and shape a clear, hypothesis-driven project aligned with your interests and available resources.
Suggested timeline for completing milestones
| Phase / Timing | Milestones |
|---|
| Phase 1 – Mid 1st Year | Develop research idea; complete literature review; formulate hypothesis; complete study design modules and assessments |
| Phase 2 – End 1st Year | Finalize methodology; complete CITI training; submit IRB |
| Phase 3 – Mid 2nd Year | Complete data collection; complete core statistics training; perform data analysis |
| Phase 4 – End 2nd Year | Present at a national conference; draft manuscript |
| Phase 5 – End 3rd Year | Submit manuscript for publication; revise as needed following peer review |
| Phase 6 – End 3rd Year | Post-publication engagement; understand and track the broader academic impact of the work |
Evaluations will be done by the Research Director and the PI/mentor of the project. Evaluations will largely be based on completion of milestones. Milestones will depend on the current progress and year of fellow so will be specified at the beginning of each research rotation.
Milestones and tasks will be accessible in the Pulm research database site (login at hfhpulm.com)
Preliminary requirements to be done before starting the research track.
1. Citi Training (Research Ethics and Compliance Training). This is required for all individuals engaging in research at Henry Ford. Email Jeff Jennings and Jackie Day with the certificate.
2. End note installation. Endnote integrates into Word and is used to track references for your manuscript
3. Make sure you have a login for the research database at hfhpulm.com (direct link: http://rhwweb392c/telephone/main_login.php). Kesha will have sent you an email with your login information.
You can look at all of the active projects that are ongoing as well so as to help choose a mentor for your research track.
Primary Research Project By January of 1st year, each fellow should have a working idea and a hypothesis. This will be a process that is done in conjunction with your staff mentor. This process involves a simple checklist:
- Has it been done before?
- Is the question important/pertinent; does it introduce new knowledge on the subject (the “who cares” test)?
- Is it feasible?
The project or even idea will likely need to be modified (or rejected altogether) as the question fails one or more of these checklist questions.
In mid-January the idea should be presented to the pulmonary group. An interested PI may be assigned on that day unless the project already has a PI.
You should expect to start the project after IRB approval and present your findings at a national conference, sometime before you graduate.
Case Reports do qualify for the scholarly activity requirement for your fellowship. However, they MUST be presented nationally and written up (in publishable form) before graduation.
Quality initiatives These projects are very similar to the original research project. They can still be published. The difference is you are testing a process. With a research project you are testing a hypothesis.
QI Process: How can we implement a process to improve compliance with ventilator weaning?
Research: Hypothesis: a ventilator bundle team increases compliance with vent weaning, as compared to a control group.
Guidelines for authorship
First Author
By default, the fellow who initiates a de novo original research project will be first author. Maintaining first authorship requires active and sustained involvement throughout the project, including participation in study design, IRB submission, data collection, data analysis, and manuscript preparation.
Mentor involvement at any stage does not diminish the fellow’s first authorship, as active engagement by the PI is expected in scholarly work.
Senior Author (Corresponding Author)
The staff Principal Investigator (PI) will serve as senior author and corresponding author. This is typically the last author listed on the manuscript and reflects overall supervision and responsibility for the project.
Middle Authors
Individuals who contribute in a meaningful way to the project should be considered for middle authorship. Contributions may include manuscript writing, statistical analysis, data collection, study design input, or other substantive components of the work.
Authorship order should reflect relative contribution. In general, the greater the contribution, the higher the placement among the middle authors (e.g., a second author has typically contributed more than a third author). While contribution involves some judgment, placement should align with the scope and impact of each individual’s work.
Additional staff, fellows, or residents who join a project will be assigned authorship based on their level of involvement. For example, someone working closely alongside the senior fellow throughout the project may merit second authorship.
If the original fellow reduces participation substantially or does not contribute meaningfully to manuscript preparation, they may remain an author if they made a significant contribution; however, first authorship may be reassigned if another individual ultimately contributes more to completing the project.
Writing the Introduction for an Original Research Study
Materials
- PowerPoint lecture
- Literature review module: https://alison.com/topic/learn/86305/literature-review
Pre-Requirement
EndNote must be installed prior to beginning this assignment.
Assignment
Write an introduction that builds logically and directly toward a clear, testable hypothesis.
This may or may not become your primary research project. That is not the objective. The purpose of this early exercise is to:
- Practice structured literature searching
- Incorporate references appropriately using EndNote
- Construct a logical scientific argument
The introduction should move from broad background (Point A) to a specific hypothesis (Point B).
The goal is not to summarize everything known about a topic. The goal is to justify your hypothesis.
Structure of the Introduction
Each section should become progressively more specific:
- State the importance of the overall problem
(Why does this matter clinically or scientifically?) - Summarize what is currently known
Support statements with key references. - Identify gaps or unanswered questions
What remains unclear? What has not been adequately studied? - Provide a logical rationale
Offer a plausible explanation or mechanism that addresses the gap. - State your hypothesis
The final sentence of the introduction should clearly state the specific hypothesis your study will test.
Key Principle
You should know your hypothesis before writing the introduction.
The introduction is constructed to justify it.
If the hypothesis does not feel like a natural conclusion of the preceding paragraphs, revise the introduction.
Pre-Requirements: EndNote should have been installed.
Your assignment is to write an introduction that leads up to a hypothesis. NOTE: this may or may not end up being your actual project. If it is, that’s nice but the point of this exercise early in your elective is to practice searching for literature and incorporating references into the introduction. The goal of the introduction is to get from point A (background) to point B (the hypothesis).
In other words, the purpose of an introduction is to justify the hypothesis of your project. This is done in 4 steps. Each step gets more SPECIFIC towards the ultimate hypothesis
1. state the importance of the overall problem (eg. COPD is the leading cause of death in smokers)
2. State what is known about this problem (references)
3. State what holes or unanswered questions remain about the problem
4. State a potential reason for this
5. State your hypothesis which is attempting to test one of those unanswered questions in step 3.
In order to write the intro, you would of course already have the hypothesis and then you work toward that (hypothesis is usually the last sentence in the introduction).
Scientific Method
The scientific method is a process whereby you making conjectures (hypotheses) and derive predictions from them. You then carrying out experiments or empirical observations based on those predictions. When you start a project, you will be in essence following the principles of the scientific method.
The steps of the scientific method:
1. Make an observation or observations.
Example: Perhaps on rounds a question would arise We sure are ordering a lot of EEGs for encephalopathy.
2. Ask questions about the observations and gather information (this is the literature or background review).
How useful is the EEG…i.e. what is the diagnostic yield in the medical ICU??
3. Form a hypothesis — a tentative description of what’s been observed, and make predictions based on that hypothesis.
“EEGs increases frequency of hospice consults in patients with unspecified encephalopathy”
Generic: “If _____[I do this] _____, then _____[this]_____ will happen.”
4. Test the hypothesis and predictions in an experiment that can be reproduced.
-This is where study design comes into play. Choose the setting (patient population) and the type of study to test the hypothesis (RCT, case-control, etc).
-this step will also involve the ultimate collection of the data (either prospectively or via chart review).
5. Analyze the data and draw conclusions; accept or reject the hypothesis or modify the hypothesis if necessary.
6. Reproduce the experiment until there are no discrepancies between observations and theory.
“Future studies are needed to confirm our findings of this single center non-randomized observational trial”
7. Communicate Your Results
Presentation at national conference and then publication of the manuscript.
Some key underpinnings to the scientific method:
- The hypothesis must be testable and falsifiable Falsifiable means that there must be a possible negative answer to the hypothesis.
- Research must involve deductive reasoning and inductive reasoning. Deductive reasoning is the process of using true premises to reach a logical true conclusion while inductive reasoning takes the opposite approach.
- An experiment should include a dependent variable (which does not change) and an independent variable (which does change).
- An experiment should include an experimental group and a control group. The control group is what the experimental group is compared against.
Conclusions
Assignments:
– scientific method work sheet Sponge Bob
Study Designs
Choosign the correct study design is critical for successfully testing your hypothesis. A hypothesis can be test experimnentaly (you provide an intervention and compare to a control group) or by observation “you observe the phenomena without intervening, and comapre to a natural (control) group.
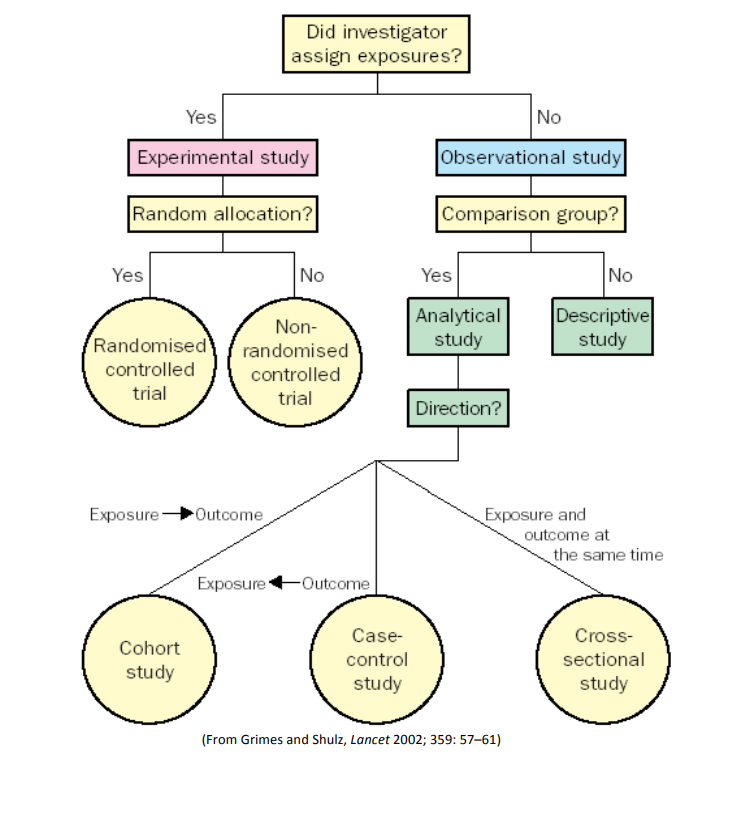
Please refer to this handout for a description of the study designs.
Special mention: Pilot and Feasibility Trials
Tests feasibility for a larger trial (outcomes are things like recruitment rates, retention rates, completion of action plans, completeness of data) with those issues checked off, the larger study can have the official outcomes to test the hypothesis. Feasibility trials are not really for “hypothesis generating” etc.
https://youtu.be/bljPT9whkQo (Pilot vs feasibility 25:46 mark of 1:01:35)
Handout: BMC Medical Research Methodology 2010:10:1
Topic 5 Power analysis.
All studies that have a comparison (control) group require determination of sample size. This is the minimal number of subjects needed to provide enough “power” adequately compare the control and the experimental group and accept or reject your null hypothesis. In English – how many subjects do you need to test your hypothesis. If the two groups show no difference for a given outcome, how many subjects do you need to be able to say that this is truly no difference? If you have 5 patients in each group and 4 die in only one group, is that because you have too low of a sample size?
In determining a sample size, you need to know the prevalence of the disease in that population you are studying AND you need to say what you think the difference would be between groups.
Videos:
1. Statistical Power, Clearly Explained https://www.youtube.com/watch?v=Rsc5znwR5FA
2. p values clearly explained https://www.youtube.com/watch?v=5Z9OIYA8He8
3. Sample size table – https://www.research-advisors.com/tools/SampleSize.htm
4. Webinar Series: Sample Size https://www.youtube.com/watch?v=PZcm57CTKsk&feature=youtu.be
Topic 8 How to write a manuscript
Readings:
How to write a scientific manuscript for publication – Blood Transfus. 2013 Apr; 11(2): 217–226.
Other articles located in the curriculum folder.
